 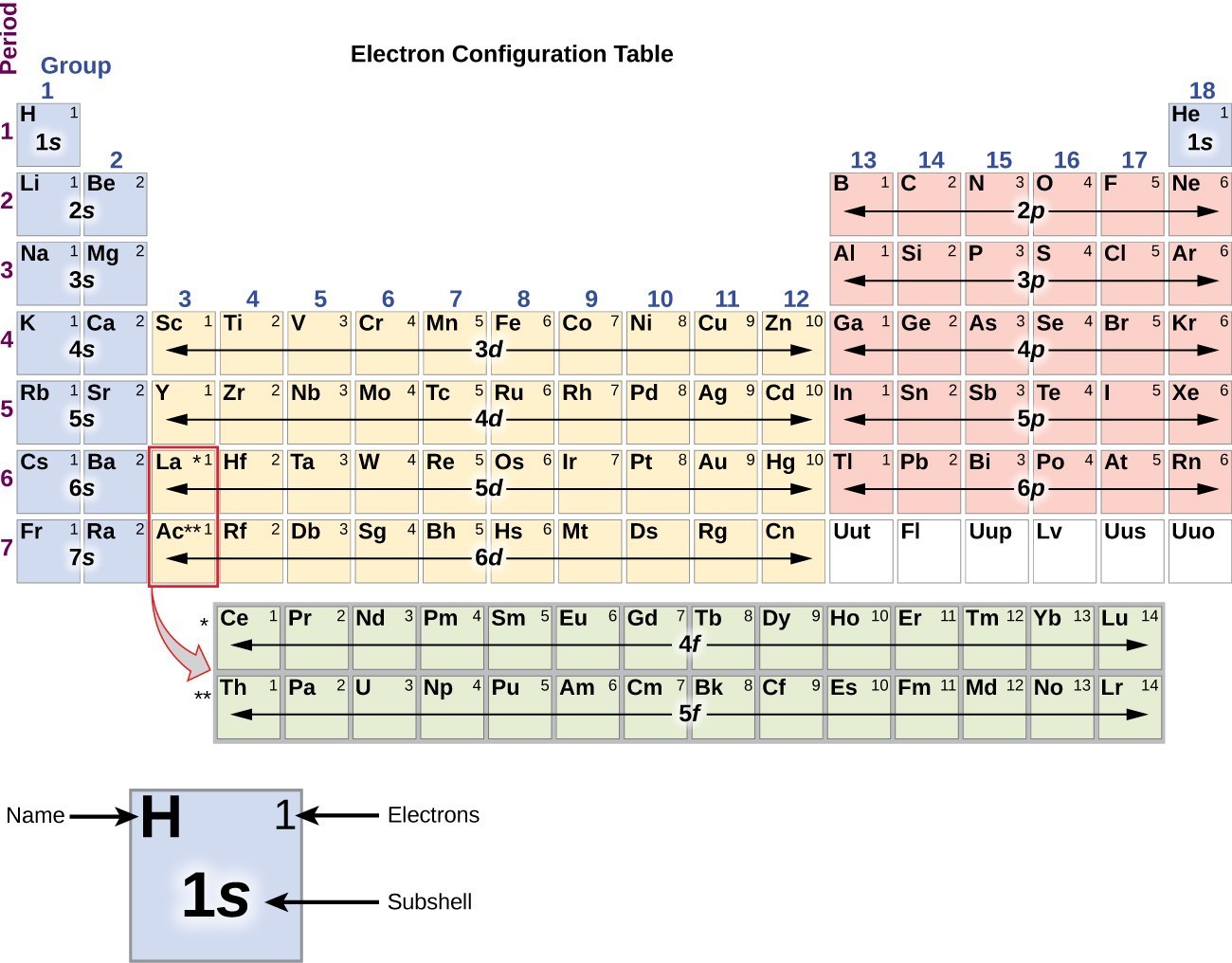
Valence electron configuration is a fascinating aspect of chemistry that provides valuable insights into the periodic table and its trends. By studying the valence electron configuration, scientists can predict an element’s behavior and its ability to form compounds. The arrangement of valence electrons determines an element’s reactivity, chemical bonding, and overall stability. In conclusion, understanding valence electron configuration is crucial in comprehending the behavior and properties of elements. Unlike main group elements, transition metals have variable valence electrons, meaning they can lose different numbers of electrons and form multiple positive charges. Transition Metals Have Variable Valence Electrons The octet rule states that atoms tend to gain, lose, or share electrons in order to achieve a stable electron configuration with eight electrons in the valence shell, resembling the electron configuration of noble gases. 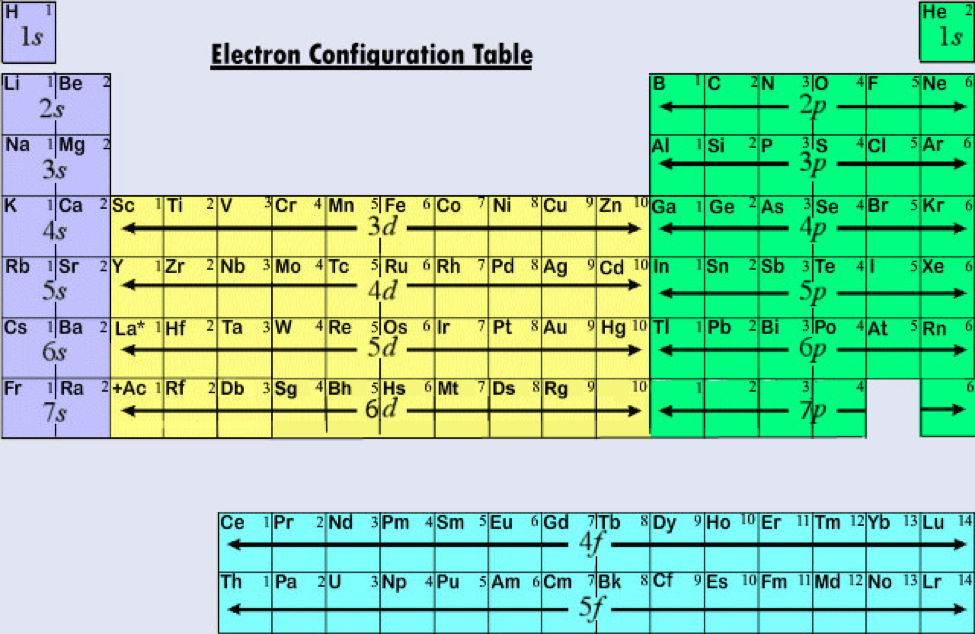
Read also: 9 Mindblowing Facts About EnzymeSubstrate Complex Valence Electrons Follow the Octet Rule Elements with the same number of valence electrons are grouped together. The number of valence electrons determines an element’s position in the periodic table and its chemical behavior. Valence Electrons Determine the Periodic Table Their interaction determines how atoms combine to form molecules. Valence electrons play a crucial role in the bonding of atoms and the formation of chemical compounds. It determines the chemical properties and reactivity of an element. Valence electron configuration refers to the distribution of electrons in the outermost energy level, also known as the valence shell, of an atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
