 
There is no other tool in science that allows us to judge relative properties of a class of objects like this, which makes the periodic table a very useful tool. The variation of properties versus position on the periodic table is called periodic trends. One of the reasons the periodic table is so useful is because its structure allows us to qualitatively determine how some properties of the elements vary versus their position on the periodic table. Be able to state how certain properties of atoms vary based on their relative position on the periodic table. See the site’s content usage guidelines.1. 
The graphic in this article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. This graphic can be downloaded as a PDF below, and is also available to purchase as large posters, or on a mug, on the site’s Redbubble page.Īlso, check out the Compound Interest Periodic Table of Data, which visualises these trends and more! Both of these factors can lead to lower than expected first ionisation energies. Paired electrons in the same orbital can lead to repulsion, again making an electron easier to remove. Electrons in p orbitals are slightly easier to remove than those in s orbitals of the same energy level. The first ionisation energy generally increases from left to right across a period, as the electron is drawn closer to the nucleus by the increased nuclear charge and becomes harder to remove. This increase in electronegativity across a period is due to the increased nuclear charge and approximately constant shielding effects resulting in a greater force of attraction to the nucleus of the atom felt by the bonding electrons. Generally, electronegativity increases moving towards the top right of the Periodic Table. Strength of these is determined by the size of the molecule.Įlectronegativity is a measure of the tendency of an atom to attract a bonding pair of electrons. Simple molecular substances have low melting points as only weak intermolecular forces must be overcome in order to melt them. The majority of non-metals have a simple molecular structure. For both, this is due to the fact that the bonds require a lot of energy to break. 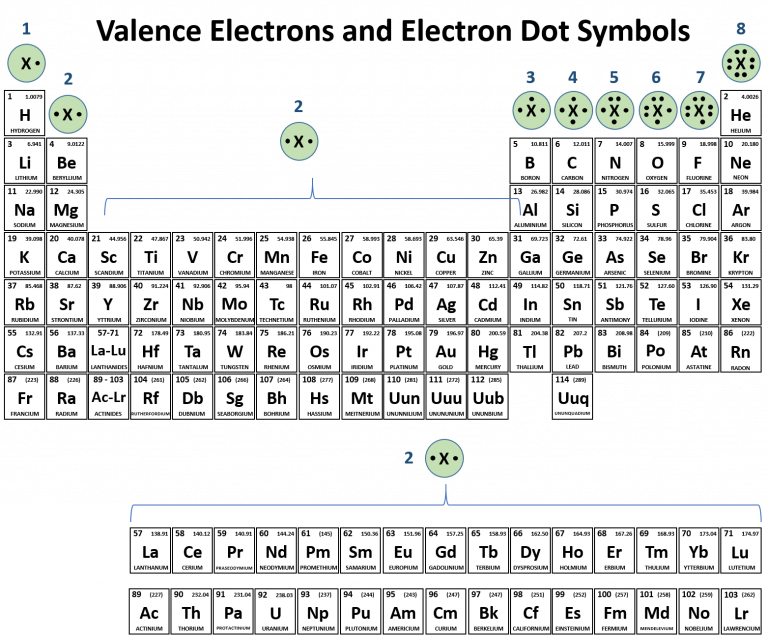
Metallic bonded and macromolecular substances tend to have high melting points. This leads to a increase in atomic radius despite the increasing nuclear charge down a group. Atomic radius increases down a group as valence electrons become increasingly distant from the nucleus, and shielding also increases. Each diagram is also accompanied by a brief explanation of the trends, and a simple explanation of basic anomalies.Ītomic radius decreases across a period as nuclear charge increases but shielding effects remain approximately constant, resulting in electrons being drawn closer to the nucleus. The emphasis of this graphic is on giving a general impression of each of the trends although a rough scale is given to the right of each diagram, this is intended to be for approximate reference, rather than allowing the diagrams to be used as precise tools for determining properties of specific elements. These properties show trends, also referred to as ‘periodicity’, as you progress along a period. This graphic looks at several properties of elements in the Periodic Table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
